Feb
17
2022
On demand
AAV manufacturing process development using fast USP and DSP HPLC analytics
Thursday 08:00 PST / 11:00 EST / 16:00 GMT / 17:00 CET
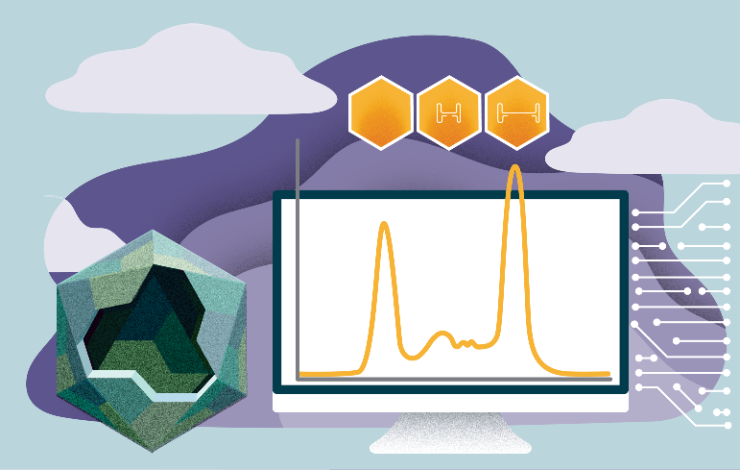
One of the key challenges in manufacturing viral vectors is to increase the ratio between empty and full capsids.
When the expression in the cell line results in less than 10% full capsid it is quite impossible to reach better than 90% full in the final product. It is therefore mandatory to optimise the USP to result in better empty/full ratio.
This can be efficiently realised by using at-line HPLC to allow for analysis of the full and empty capsids ratio directly in the harvest.
The residual empty capsids can be removed by polishing step using different anion exchange columns.
- AAV is the leading vector in the field of gene therapy and it is therefore crucial to develop a robust and high efficiency platform for its manufacturing.
- In-process analytics of vector capsid production is a critical optimization target in development of AAV-based gene therapy products.
- In this presentation we introduce a fast at-line HPLC based system that enables analysis of the full and empty capsids ratio directly in the harvest.
- Insights about purification will also be discussed.
You might also like

Same-section spatial multiomics: a platform for detailed analysis of the solid tumor TME
Emily Neil, Ariadna Pascual
27 June
in 71
Days
Pure and simple: understanding LNP analytics for better mRNA-based drugs
Adam Crowe
29 May
in 42
Days

Empowering patient care: in-process and quality control solutions for cellular therapies
Christin Tischner, Simon Lennartz
23 May
in 36
Days

T cells from beginning to end: optimizing leukopak and drug product cryopreservation
Meredith Safford, ShaNelle Yelity
22 May
in 35
Days
Mastering AAV production: best practices For small- to large-scale manufacturing success and reduced development time
Emily Jackson-Holmes
21 May
in 34
Days

