The comprehensive characterization of critical quality attributes (CQAs) for the safety and efficacy of the viral vector material produced for gene therapy trials remains a key challenge for the advanced therapies sector. Viral vector analytical development teams are required to address issues such as identification, characterization, and enumeration of undesired contaminants such as mycoplasma.
With the CMC-related requirements of the US FDA, EMA, and other major regulatory agencies still evolving and steadily increasing in stringency, it is more important than ever to harness the cutting edge in analytical tools to improve the identification and measurement of these CQAs.
Fortunately, a new generation of analytical tools designed to meet the specific needs of viral vector production is arriving, offering the enhanced speed, sensitivity, and robustness that industry requires.
The following curated collection of content offers technical and strategic insights and practical advice relating to:
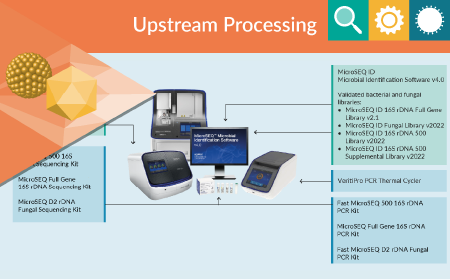
 Analytical development
Analytical development Cell therapy manufacturing
Cell therapy manufacturing Expression systems
Expression systems Mycoplasma testing
Mycoplasma testing Regulatory CMC
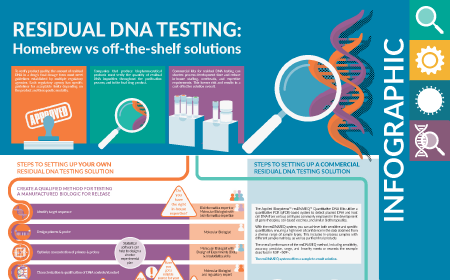
Regulatory CMC Residual DNA testing
Residual DNA testing Vector production
Vector production